More than ever, patients and caregivers are turning to social media to share their experiences and treatment journeys, seek advice, or discuss pharma and healthcare brands. In light of this shift to the online realm, how can the pharmaceutical industry harness online conversational data to improve patient outcomes and inform strategic decision-making?
In recent years, the healthcare industry has decreased its reliance on clinical trial data in favour of Real World Evidence (RWE). But in spite of the ever-growing popularity of this established research method, there is still hesitance about ‘Social RWE’ – the data that can be mined from online conversations. While there are legitimate concerns regarding ambiguous compliance and legal requirements, steps being taken by regulatory agencies to promote the use of Social RWE indicates the tide is changing.
![]()
At least 4.5 billion people worldwide were using social media in July 2021 – over double the number of users in 2015. Combine this with online blogs, crowdsourcing platforms, search engines, and video sharing, to name but a few online information sources, the world wide web comprises colossal volumes of consumer data. This pot of gold is bursting with real-world insights ripe to help improve patient care and guide business decisions.
To leverage the benefits of Social RWE and catch up with other industries already reaping the rewards of well-executed social intelligence practices, pharma companies must first recognise the value this data offers them and their stakeholders.
Key Areas of Benefit Throughout the Clinical Pipeline
Social RWE can be exceptionally valuable at each stage of the clinical pipeline. Below we introduce the six key areas of benefit.
- Understanding the patient journey: Social RWE enables companies to identify conversational trends and emotions at different stages of the patient journey, from pre-diagnosis all the way through to treatment, management, remission, relapse, and even up to death.
- Identifying unmet needs at each stage of the patient journey: Companies can pinpoint unmet needs that would otherwise remain unknown, simply because online conversations tend to be unfiltered and authentic narratives of a patient’s journey. Emotions and sentiments can be mapped to these unmet needs, illuminating the reality of life with a specific medical condition.
- Evaluating real world outcomes: Patient-reported outcomes (PROs) are now routinely incorporated into clinical trials. However, they are typically collected using questionnaires or surveys that do not fully capture the experience of patients. Online conversations are unstructured, reflecting the natural tones, sentiments and emotions of patients and providing a 360-degree view of their experience.
- Building enhanced value propositions: By analysing large and detailed datasets, Social RWE can reveal drug benefits that were not detected during traditional clinical trials. Indeed, this data can help pharma go beyond safety and efficacy to really understand the impact of a treatment
- Accelerated drug development and achieving market success: A deep-dive into social conversations can help create detailed portfolios of key stakeholders, their experiences, needs, preferences, and topics of interest. Such insights are critical to personalised marketing strategies that achieve maximum engagement.
- Pharmacovigilance: Adverse effects often remain undetected and under-reported in routine clinical care. Owing to the anonymity of the internet, patients are more likely to be honest and share concerns regarding their symptoms, side-effects and QoL events. This enables a more robust capture of adverse effects to help enhance quality control and improve treatment experiences.
The key challenge faced by companies wishing to reap these benefits is identifying quality data from the noise. We use a two-tiered model to achieve this:
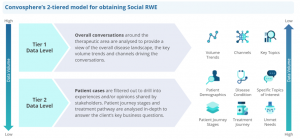
- Tier 1: Overall conversations around the therapeutic area are analysed to provide a view of the overall disease landscape, the key volume trends and channels driving conversations.
- Tier 2: First-person conversations are filtered out to drill down into mentions and content shared by stakeholders (patients, caregivers, HCPs, PSGs or organisations). Conversations are analysed in-depth to gain a detailed understanding of the experiences, opinions, patient journey stages as well as treatment specific concerns.
Explore the Value of Social RWE in our new whitepaper
To learn more about this two-tiered model and how pharma can leverage Social RWE to achieve patient-centred business imperatives, download our whitepaper Optimising Real-World Evidence with Big Social Data. Key topics discussed in the whitepaper include:
- What is RWD in the context of Big Social Data?
- What is the difference between traditional social listening and RWE social listening?
- What RWD exists in Big Social Data?
- Why does RWD from social listening matter?
- What is the value of social RWD throughout the clinical pipeline?
- What are the challenges of RWE from Big Social Data?
To learn more about how Convosphere can help you gain insight into the patient experience by harnessing real-world evidence from big social data, contact us today.




